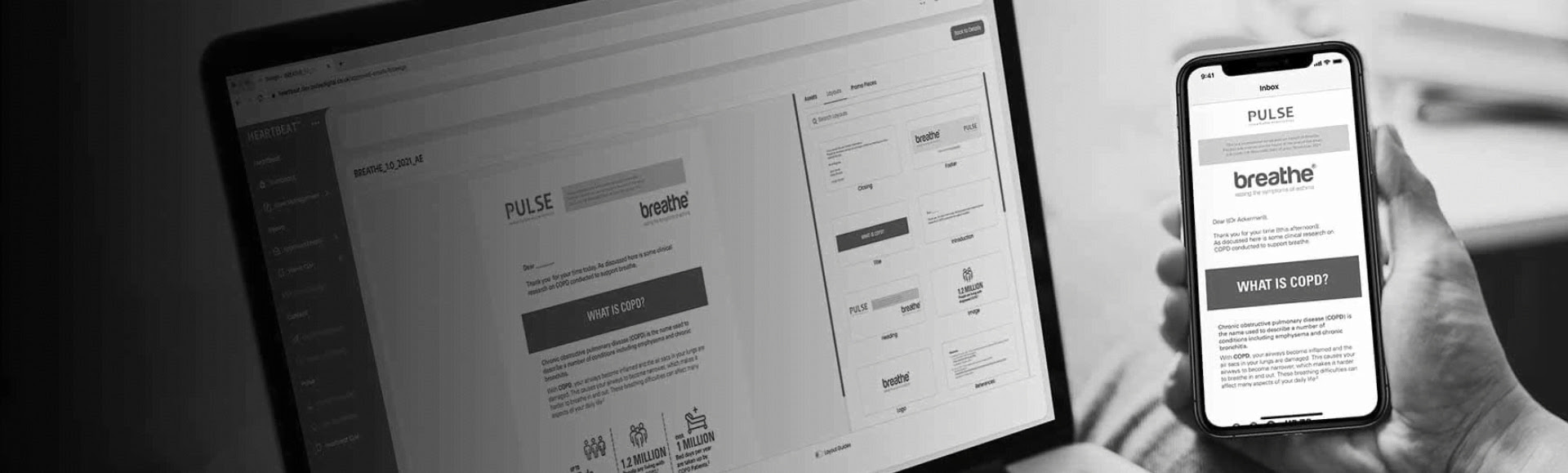
What is Veeva Approved Email?
Veeva Approved Email (VAE) is a specialised email communication tool designed for the pharmaceutical and life sciences industries. It ensures that all emails sent to healthcare professionals (HCPs) are compliant with industry regulations, personalised, and effective. This platform streamlines the creation, approval, and distribution of emails, helping pharma brands maintain regulatory compliance while enhancing engagement with their audience.
Key Features and Functionalities of Veeva Approved Email
Discover the essential features and functions of Veeva Approved Email for streamlined communication and compliance in the healthcare industry.
-
Pre-Approved Email Templates
Ensures that all emails meet regulatory standards and maintain brand consistency.
-
Email Fragments and Template Fragments
Allows for the creation of reusable content blocks and design elements, speeding up the email creation process.
-
Personalisation
Leverages Veeva CRM data to tailor emails to the specific interests and needs of individual HCPs, enhancing relevance and engagement.
-
Shared Documents
Provides easy access to compliant and up-to-date resources such as PDFs and brochures, which can be linked or attached to emails.
-
Compliance Management
Tracks and documents consent preferences to ensure that communications are only sent to those who have opted in, maintaining adherence to data privacy regulations.
-
Analytics and Reporting
Detailed insights into engagement metrics, such as open rates and click-through rates, allowing for continuous optimisation of email campaigns.
-
Content Approval Workflow Integration
Through integration into Veeva Vault, streamlines the MLR review, approval, and dissemination of approved assets, reducing the time and effort required to manage email communications.
Benefits of Using Veeva Approved Email

Ensuring Compliance with Industry Regulations
Veeva Approved Email ensures that all communications with healthcare professionals (HCPs) adhere to stringent industry regulations. In the highly regulated pharmaceutical and life sciences industries, maintaining compliance is paramount to avoid legal repercussions and maintain a reputable standing. Veeva Approved Email streamlines this process by providing built-in compliance features.
It ensures that emails sent to HCPs are pre-approved, reducing the risk of unauthorised or non-compliant content being disseminated. This tool also tracks all communications, offering an auditable trail that meets regulatory requirements such as those outlined by the FDA, EMA, and other global regulatory bodies. By using Veeva Approved Email, organisations can confidently communicate within the legal frameworks and avoid costly fines or penalties.

Personalising Communication with HCPs
Veeva Approved Email allows for highly personalised communication with HCPs, which is crucial for building and maintaining strong professional relationships. Personalisation can include tailoring email content to reflect the specific interests, needs, and preferences of individual HCPs. This can be achieved through the integration of CRM data, which provides insights into the HCPs' past interactions, specialties, and prescribing behaviors.
By delivering relevant and personalised content, Veeva Approved Email helps in making communications more meaningful and effective. Personalised emails can significantly increase the likelihood of engagement, as HCPs are more inclined to respond to communications that resonate with their specific professional interests and requirements.

Enhancing Engagement and Tracking Metrics
Veeva Approved Email offers advanced features to enhance engagement and track key metrics. Engagement can be significantly improved through the use of interactive content, targeted messaging, and timely follow-ups, all of which are facilitated by Veeva’s robust platform. The system also provides detailed analytics and reporting capabilities, allowing organisations to monitor open rates, click-through rates, and other vital metrics.
These insights enable marketers and sales representatives to understand the effectiveness of their campaigns and adjust strategies in real-time for better outcomes. The ability to track and analyse engagement metrics helps in fine-tuning communication efforts, ensuring that the messages are not only delivered but also read and acted upon.

Improving Productivity and Efficiency
Veeva Approved Email enhances productivity and efficiency by streamlining the communication process between organisations and HCPs. The platform's user-friendly interface and integration with Veeva CRM allow for the quick creation, approval, and dissemination of emails. Automated workflows reduce the time and effort required to manage email campaigns, freeing up valuable resources that can be redirected towards other critical tasks.
Additionally, the centralised repository of pre-approved email templates ensures that sales representatives and marketers can rapidly deploy consistent and compliant messages without the need for repeated approval cycles. This efficiency not only speeds up communication but also ensures that HCPs receive timely and relevant information, ultimately leading to better engagement and stronger professional relationships.
How Veeva Approved Email Works
Veeva Approved Email simplifies and enhances your communication with healthcare professionals (HCPs) by ensuring every email you send is compliant, personalised, and impactful. The platform integrates seamlessly with Veeva CRM, allowing you to quickly access and use pre-approved email templates that are both compliant with industry regulations and tailored to meet the specific needs of your audience.
With Veeva Approved Email, you can personalise your messages based on detailed insights into HCPs' preferences and past interactions. It also provides robust tracking and analytics, enabling you to monitor engagement and effectiveness, so you can adjust your strategies in real-time. Ultimately, Veeva Approved Email streamlines your communication efforts, ensuring you can reach your audience with the right message at the right time, while maintaining regulatory compliance and boosting productivity.
Key Components of Veeva Approved Email
Discover the essential features and functions of Veeva Approved Email for streamlined communication and compliance in the healthcare industry.
-
Email Templates
Email templates are the foundation of Veeva Approved Email, providing pre-designed and compliant structures for communication.
These templates are created to ensure consistency and regulatory adherence across all emails sent to healthcare professionals (HCPs).
They can be customised with specific content tailored to the recipient's needs, ensuring that each email is both relevant and engaging.
-
Email Fragments
Email fragments are reusable content blocks within the templates.
These can include standard messages, disclaimers, and other commonly used text or multimedia elements.
By utilising email fragments, brand managers can quickly assemble emails that are consistent with brand guidelines and regulatory requirements, while also saving time in the content creation process.
-
Template Fragments
Template fragments are components of the email templates that can be shared across different templates.
These fragments might include header or footer sections, call-to-action buttons, or other design elements that need to remain consistent across multiple email campaigns.
Using template fragments ensures a uniform look and feel, enhancing brand consistency.
-
Shared Documents
Shared documents are resources like PDFs, brochures, and detailed product information that can be linked or attached to emails.
These documents are stored centrally, ensuring they are up-to-date and compliant.
This feature allows brand managers to provide HCPs with additional information that supports the email’s message without needing to include extensive text within the email itself.
Systems Involved
Veeva Approved Email integrates with a number of other systems and technologies
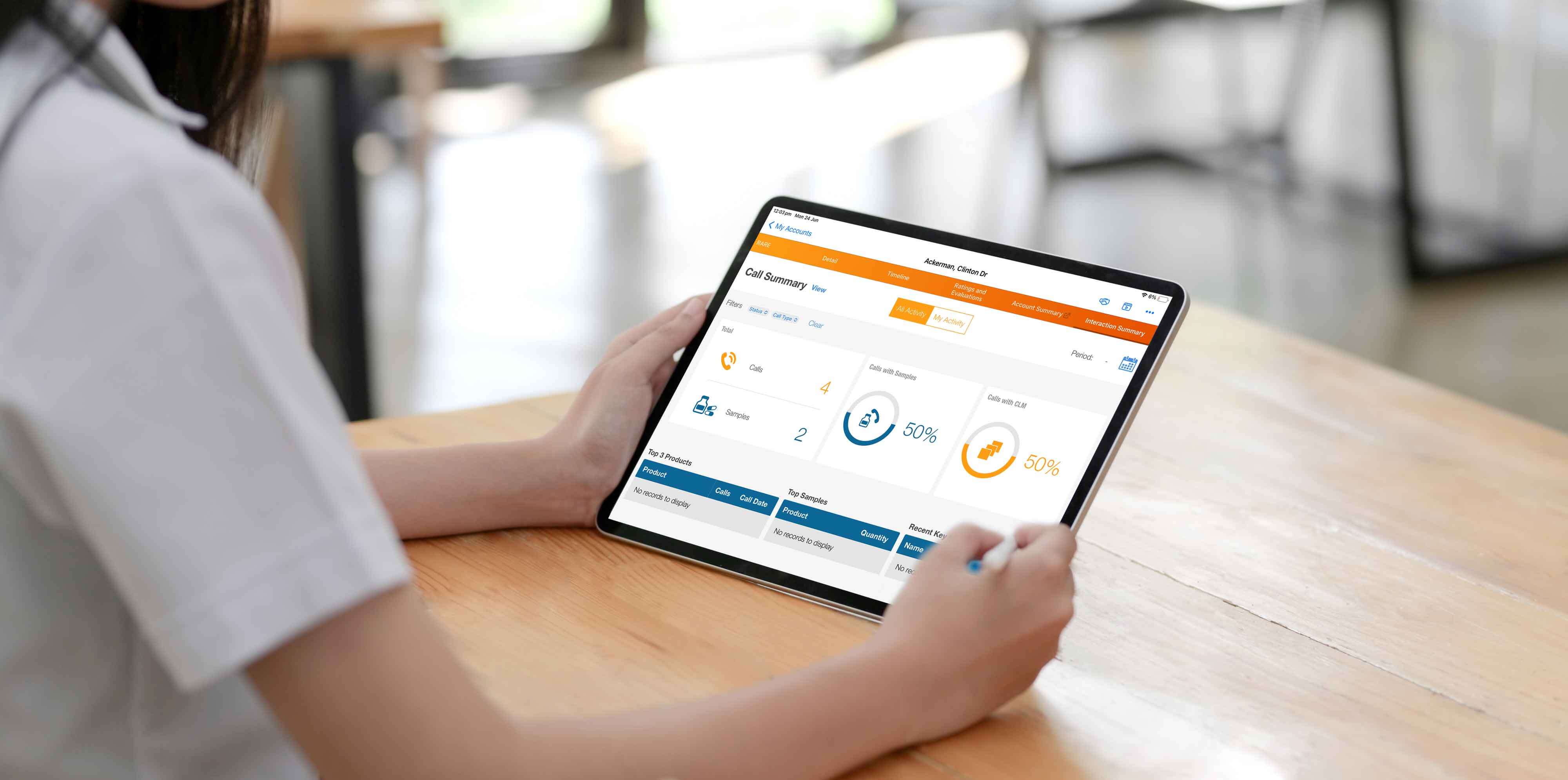
Veeva CRM
Veeva CRM is the central system that manages customer interactions and data.
It integrates seamlessly with Veeva Approved Email, allowing brand managers to access detailed insights about HCPs.
This integration enables the personalisation of emails based on past interactions, preferences, and behaviors, ensuring more targeted and effective communication.

Veeva Vault
Veeva Vault is the content management system where all the email templates, fragments, and shared documents are stored and managed.
It ensures that all content used in email communications is compliant with industry regulations and up-to-date.
The integration of Veeva Vault with Veeva Approved Email streamlines the content approval process, reducing the time required to get emails out to HCPs.
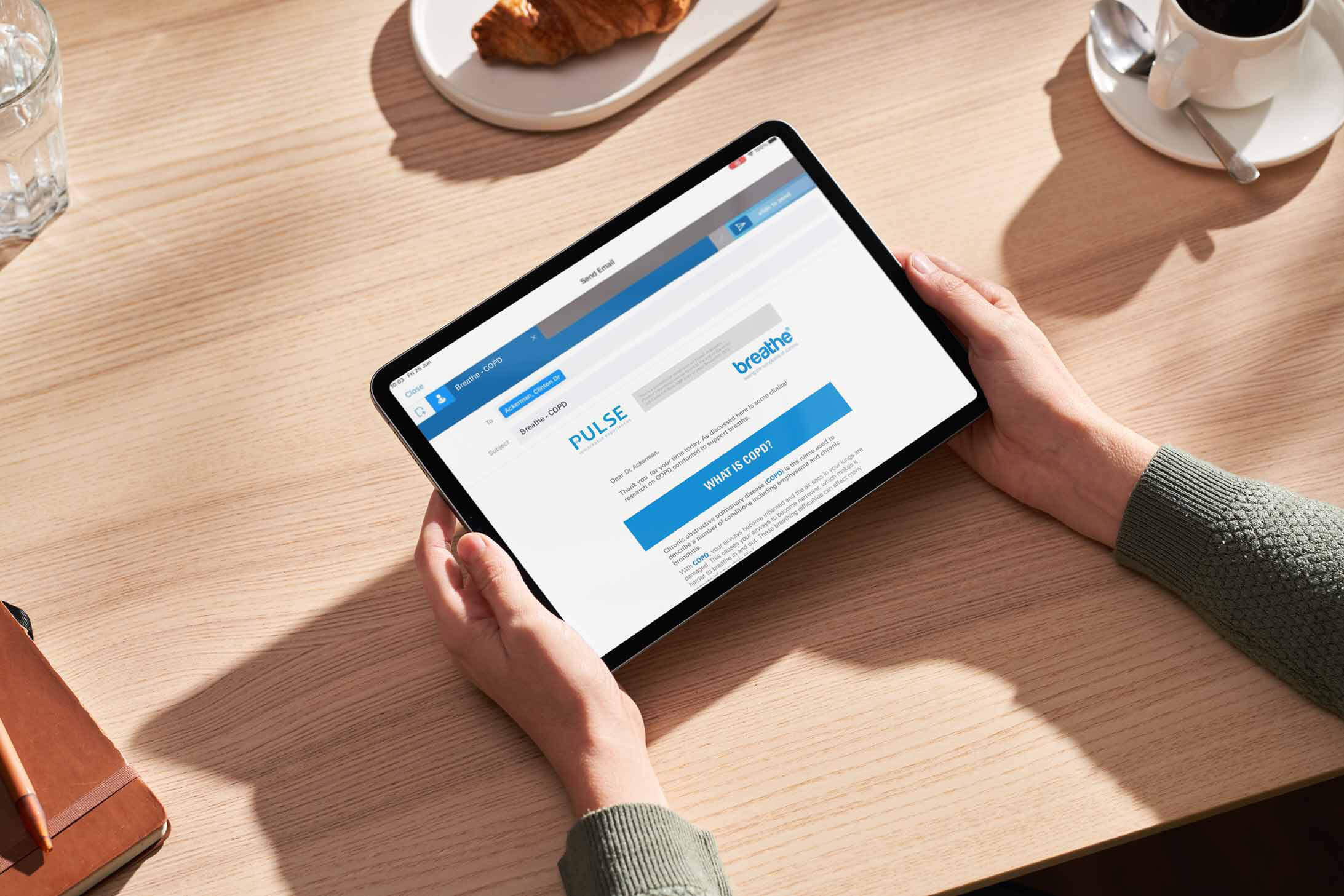
Enterprise Email Engine
The enterprise email engine is the system responsible for the actual sending of emails.
It handles the delivery of emails to the intended recipients, ensuring high deliverability rates and managing bounce-backs or delivery failures.
This engine is crucial for ensuring that emails are sent efficiently and reliably, reaching HCPs as intended.
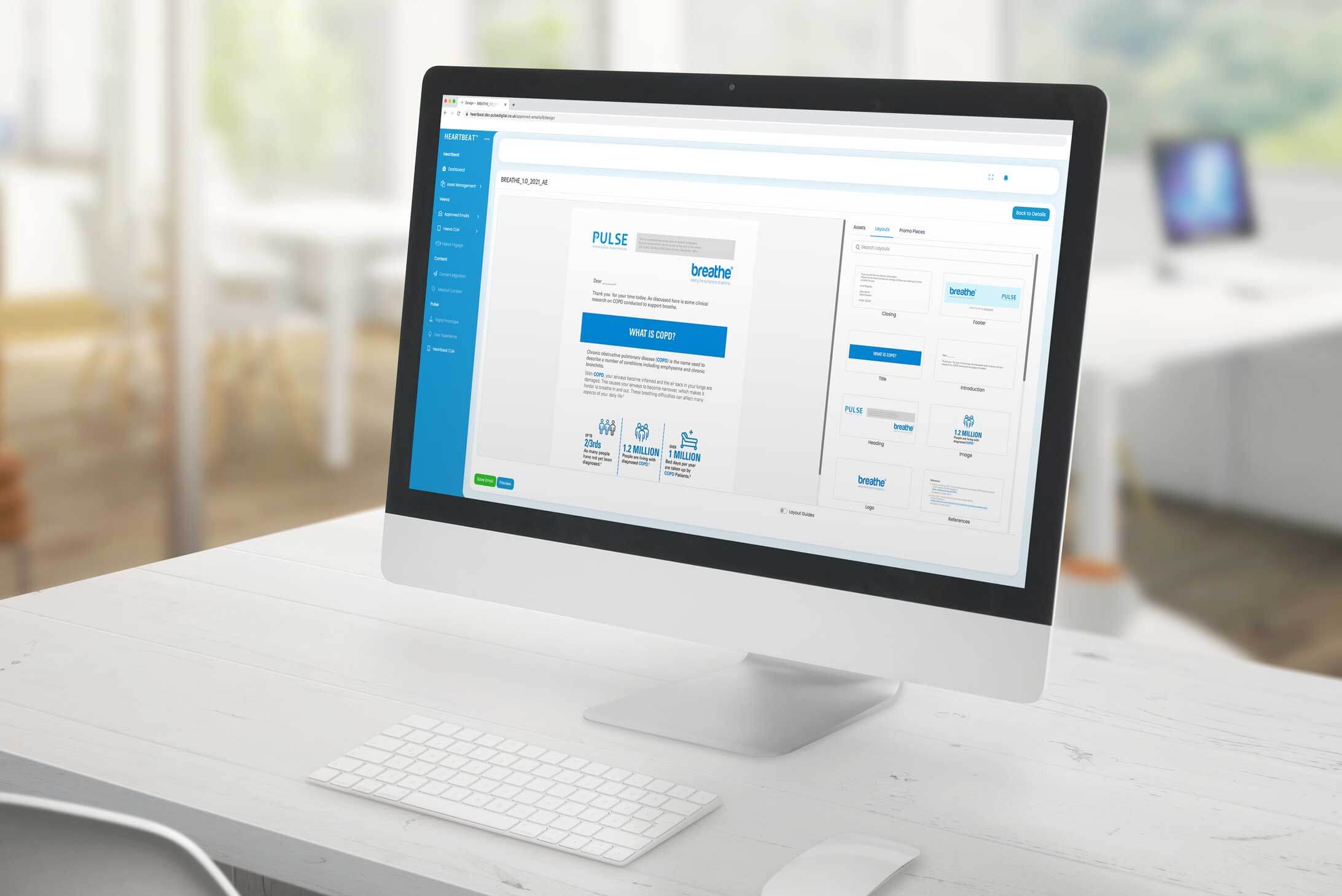
HeartbeatTM Veeva Content Authoring System
The HeartbeatTM Veeva Content Authoring System is a tool used to create and manage the various components of Veeva Approved Email, including templates, fragments, and shared documents.
It provides an intuitive interface for brand managers to design and approve content, ensuring that all elements are compliant before being used in email communications.
By streamlining the content creation and approval process, the Heartbeat system plays a vital role in enhancing the efficiency and effectiveness of Veeva Approved Email.
Approved Email Consent and Settings
Veeva Approved Email aids pharma brands in managing consent by integrating seamlessly with Veeva CRM to track and document the consent status of healthcare professionals (HCPs). This ensures that all communications are sent only to those who have explicitly agreed to receive them, adhering to strict data privacy regulations such as GDPR and HIPAA.
The platform automates the management of consent forms and preferences, providing an auditable trail of compliance. By keeping detailed records of HCPs' consent statuses and automatically updating these as needed, Veeva Approved Email helps pharma brands maintain trust and legal compliance, while effectively managing communication preferences and permissions.
Getting Started with Veeva Approved Email
Implementing Veeva Approved Email involves several key steps that ensure a smooth integration and effective use of the platform for compliant and impactful communication. Here's a detailed guide for pharma brand managers:
-
Assess Needs and Objectives
First, identify the specific needs and objectives of your communication strategy. Determine the goals you want to achieve with Veeva Approved Email, such as improving engagement, ensuring compliance, or enhancing personalisation. This will guide the customisation and implementation process.
-
Secure Stakeholder Buy-In
Engage key stakeholders, including regulatory, legal, IT, and sales teams, to ensure alignment and support for the implementation. Highlight the benefits of Veeva Approved Email, such as compliance assurance, improved efficiency, and better engagement with healthcare professionals (HCPs).
-
Training and Onboarding
Conduct comprehensive training sessions for all users who will be involved in creating, approving, and sending emails. Ensure that brand managers, sales representatives, and other relevant staff understand the features and compliance requirements of Veeva Approved Email.
-
Integrate with Veeva CRM and Veeva Vault
Ensure seamless integration with Veeva CRM and Veeva Vault. Veeva CRM will provide the necessary customer data and insights for personalised communication, while Veeva Vault will manage and store all email templates, fragments, and shared documents, ensuring they are compliant and up-to-date.
-
Create and Approve Email Templates
Develop email templates using the Heartbeat Veeva Content Authoring System. Incorporate email fragments, template fragments, and shared documents to build comprehensive and compliant templates. Ensure that all templates are reviewed and approved by the regulatory and legal teams to meet compliance standards.
-
Manage Consent and Preferences
Set up systems to manage and document HCPs' consent preferences. Integrate consent management processes within Veeva CRM to ensure that emails are only sent to recipients who have given their explicit consent. This helps maintain compliance with data privacy regulations.
-
Test and Optimise
Before fully launching, conduct thorough testing of the email system to identify and resolve any technical or compliance issues. Use pilot campaigns to gather feedback and make necessary adjustments. Ensure that tracking and analytics are set up correctly to monitor engagement and effectiveness.
-
Launch and Monitor
After successful testing, roll out Veeva Approved Email across the organisation. Continuously monitor the performance of email campaigns using the platform’s analytics tools. Track key metrics such as open rates, click-through rates, and engagement levels to evaluate the impact and optimise future communications.
-
Continuous Improvement
Regularly review and update email templates, content fragments, and shared documents to keep them relevant and compliant. Stay informed about regulatory changes and update your processes accordingly. Use insights from analytics to refine your communication strategies and improve overall effectiveness.
By following these steps, pharma brand managers can effectively implement Veeva Approved Email, ensuring compliant, personalised, and impactful communication with healthcare professionals.
Key Takeaways
Veeva Approved Email is an essential tool for pharmaceutical and life sciences companies, enabling them to communicate effectively and compliantly with healthcare professionals (HCPs). This platform integrates seamlessly with Veeva CRM and Veeva Vault, providing a robust system for creating, approving, and disseminating emails.
Its key features, including pre-approved email templates, reusable content fragments, personalised messaging, and comprehensive compliance management, ensure that all communications are not only impactful but also adhere to stringent regulatory standards.
The benefits of Veeva Approved Email are manifold. It enhances engagement by leveraging detailed CRM data to tailor communications to the specific needs and interests of HCPs. The platform's automation and workflow integration improve productivity and efficiency, allowing teams to focus on more strategic tasks. Its detailed analytics and reporting capabilities provide valuable insights into campaign performance, facilitating continuous improvement and optimisation.
Latest News & Insights

From Static to Dynamic: The Evolution of CLM eDetails in the Pharmaceutical Industry
eDetailing is the use of digital sales aids and software on tablets, laptops, and mobile devices designed to engage...

Mobile Health Apps: UX/UI Design Considerations for Developing a Mobile Health Application
Developing an effective mobile health application can be a challenging task that requires...

Accelerating and Streamlining Veeva CLM Content Creation - A Global to Local Process
The life sciences industry is experiencing a shift to hybrid engagement with HCPs which is, in turn, creating pressures on global brands...